This article was published in the September 2019 issue of Pet Food Processing. Read it and other articles from this issue in our September digital edition.
Inspections to review compliance with the Food Safety Modernization Act (FSMA) are now a reality across the animal food industry. With all compliance deadlines for the two major types of FSMA inspections — Current Good Manufacturing Practices (CGMP) and Hazard Analysis and Preventive Controls (HA/PC) — passing last September, facilities, with the exception of small facilities, which are not expected to receive HA/PC inspections until after October, are undergoing or preparing for federal inspections.
The American Feed Industry Association (AFIA) has been communicating with the US Food and Drug Administration’s (FDA) Office of Regulatory Affairs to learn how facilities are faring under federal and state inspections. In addition, AFIA has received questions from members regarding what is and is not required during inspections.
 Inspection data for FY19
Inspection data for FY19
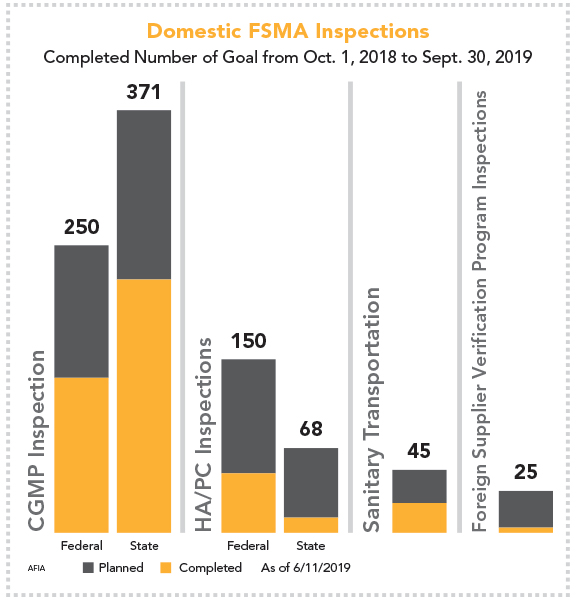
In a meeting with the FDA, AFIA discovered that, as of June 11, more than half of all planned CGMP inspections had occurred at feed and pet food manufacturing facilities across the country since Oct. 1, 2018. This included 358 inspections, carried out by federal and state inspectors, of the 621 total that are planned to take place before the end of the fiscal year on Sept. 30.
During the same time period, a little less than one-third of all HA/PC inspections had taken place, with 63 conducted of 218 planned. Only two foreign supplier verification program (FSVP) and roughly half of the sanitary transportation inspections had been conducted.
The FDA staff told AFIA it is critical that facilities keep accurate records, maintain equipment and buildings in a clean and orderly manner, and carry out effective pest control programs. Most inspector questions or concerns have fallen in these areas but, overall, the FDA said inspections had been going well considering the learning process for both state and federal inspectors on ensuring consistency in inspections, and for facilities in balancing the inspections while maintaining business operations.
Tips for FSMA inspections
AFIA members have been reaching out to the association’s legislative and regulatory staff with questions on what is expected during inspections, as well as providing general feedback on how their inspections went in terms of the questions asked, the areas the inspectors visited and any discrepancies from regulatory guidance. This information, which AFIA holds strictly confidential, allows the association to communicate generally to its members on the ways they can better ready their facilities and their staff for upcoming inspections. The following are a few reminders for pet food manufacturers:
Hazard analysis and following the plan
All animal food manufacturers must show they have completed a hazard analysis and developed a written animal food safety plan. According to the FDA, some firms are lacking an explanation in their hazard analysis on why something is not a hazard in their facility.
Inspectors have recently cited facilities for not having adequate measures in place to mitigate the risk or control the hazard. Not every hazard requires a preventive control to manage its risk. If standard operating procedures or prerequisite programs are used instead, facilities must implement and utilize those procedures and demonstrate they are being followed.
Qualified training
FSMA requires facilities to have qualified individuals with the necessary training to safely manufacture, process, pack or hold animal food appropriately. AFIA has heard inspectors are posing more questions on the Qualified Individual’s (QI) training, including the frequency and documentation. The association reminds facilities that it maintains an online video resource for individuals who need to complete this training.
FSMA also requires facilities to have employees who qualify as their site’s Preventive Controls Qualified Individual (PCQI), either through training via “standardized curriculum” or job experience. Recently, inspectors are posing more questions to verify the PCQI’s previous training meets the law’s requirements, especially when employees rely on previous work experience. Facilities can send their employees to receive a certification through the Food Safety Preventive Controls Alliance’s (FSPCA) Preventive Controls for Animal Food training, which provides the only “standardized curriculum” FDA recognizes as “adequate” for PCQI training.
Company policies for inspections
If facilities do not want photos taken during inspections, they are encouraged to formally write this in their company policies. AFIA reminds facilities that the FDA may take photos for food safety reasons and inspectors are permitted to take photos of anything they deem necessary in the interest of protecting public and animal health.
Facilities are reminded that, in general, investigators can view and copy all documents in the animal food safety plan as well as certain training records and other documents as prescribed in the FSMA final rule for animal food. Investigators generally do not have the authority to view animal food formulas or other proprietary information without justification, such as if they believe there may be a potential food safety issue.

It is important to remember that facilities may have more inspections depending on the types of pet foods they are producing or ingredients they are handling. The FDA will visit manufacturers based on their locations, previous compliance history, assigned risks or should there be due cause. The inspections may be coupled with other types of inspections and may span several days.
A good way to ensure your facility is ready for FSMA is to participate in AFIA’s Safe Feed/Safe Food Certification Program. Additionally, the FSC32 certification, for pet food and pet food ingredients, is the only program benchmarked with the Global Food Safety Initiative, meaning it meets internationally accepted standards for food safety within the food retail industry.
AFIA would love to hear from pet food manufacturers on how their inspections are going! Contact Louise Calderwood at lcalderwood@afia.org.
Read more about pet food safety on our Operations page.



